Many people take colchicine to help prevent the sudden, painful episodes known as gout flares (also known as gout attacks).1,2 The drug is available in capsule, tablet and liquid formulations.3 Some people may find that Colchicine Capsules have important advantages over the other dosage forms.4–6 Read on to learn more about 3 reasons to choose the Colchicine Capsule.4–7
Prevent gout flares
Doctors have been prescribing colchicine to prevent gout flares for centuries.8 It works by helping to prevent the redness, swelling and excruciating pain that can occur during a gout attack.9 Taking colchicine for gout flare prevention is especially important if you are just starting on a urate-lowering therapy (ULT) such as allopurinol.9 You may also want to consider colchicine if you have had gout for a while and notice that your flares are happening more frequently or becoming more severe.10
Consider colchicine capsules vs tablets
For some people, Colchicine Capsules may have advantages over tablets:
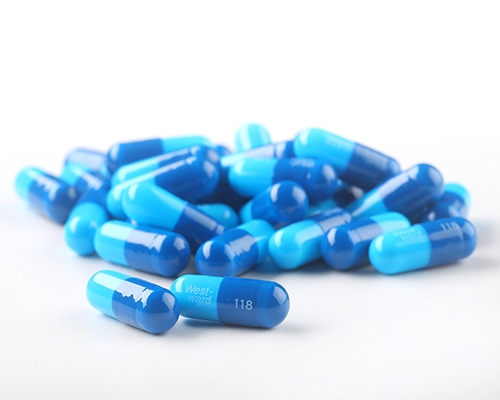
Capsules may be easy to identify
As we age, many of us need to take prescription medications each day to manage our ongoing and emerging health conditions.11 (The typical Medicare patient, for example, may take as many as two dozen different prescription drugs in a year.11) As the number of daily medications we take increases, the likelihood of medication errors may increase as well.11 Medicines that come in distinctive colors (such as the bright blue used for Mitigare® [Colchicine] 0.6mg Capsules and Generic Colchicine Capsules1) may be easier to distinguish from other medications.11,12
Capsules may be easier to swallow than tablets
For most people, swallowing a single pill isn’t a big deal. However, ease of swallowing may be important to you if you take multiple medications each day. Capsules, for example, are generally easier to swallow than tablets, given their smoother surface.4,5 In addition, the shell of a capsule is virtually tasteless, so you may be less likely to notice if the medicine inside tastes bitter or unpleasant.4,5
Capsules may save you money
If you take colchicine each day to prevent gout flares, seeing blue may help you save green. With the True Blue Savings Program, patients may pay as little as $0* for the first 30 days of Mitigare® (Colchicine) 0.6mg Capsules or Generic Colchicine 0.6mg Capsules and get refills for $5*. And Mitigare® and Generic Colchicine Capsules are covered by more than 80% of major health plans†, including:
- Humana Part D
- Cigna Part D
- Aetna Part D
- Wellcare Part D
- CVS SilverScript
- United AARP
- United Healthcare
- Express Scripts
- CVS Caremark
- Cigna Health Plans, Inc.
- Aetna
*For all eligible patients 18 years or older who are legal residents of the United States or Puerto Rico. First 30 days are as little as $0 and refills are as little as $5 for eligible patients. Maximum savings of $65 on the first fill and $50 on refills. Please see complete Terms and Conditions.
†Managed Markets Insight & Technology, LLC database and formulary status as of January 2022.
Contact your doctor
Gout is a serious medical condition that must not be ignored.13 If you have gout and suspect it may be getting worse (perhaps you are experiencing more severe flares or they are happening more often than before), it may be time for a checkup.13 Make an appointment with your doctor as soon as possible to discuss your symptoms and consider adjustments to your gout management plan, which may include the addition of colchicine therapy.2,10,13
Mitigare® is a registered trademark of Hikma Pharmaceuticals USA Inc.
Colchicine 0.6 mg capsules are contraindicated in patients with renal or hepatic impairment who are currently prescribed drugs that inhibit both P-gp and CYP3A4. Combining these dual inhibitors with colchicine in patients with renal or hepatic impairment has resulted in life-threatening or fatal colchicine toxicity. Patients with both renal and hepatic impairment should not be given Mitigare®.
Fatal overdoses have been reported with colchicine in adults and children. Keep Mitigare® out of the reach of children.
Blood dyscrasias such as myelosuppression, leukopenia, granulocytopenia, thrombocytopenia and aplastic anemia have been reported with colchicine used in therapeutic doses.
Monitor for toxicity and, if present, consider temporary interruption or discontinuation of colchicine.
Drug interaction with dual P-gp and CYP3A4 inhibitors: Co-administration of colchicine with dual P-gp and CYP3A4 inhibitors has resulted in life-threatening interactions and death.
Neuromuscular toxicity and rhabdomyolysis may occur with chronic treatment with colchicine in therapeutic doses, especially in combination with other drugs known to cause this effect. Patients with impaired renal function and elderly patients (including those with normal renal and hepatic function) are at increased risk. Consider temporary interruption or discontinuation of Mitigare®.
The most commonly reported adverse reactions with colchicine are gastrointestinal symptoms, including diarrhea, nausea, vomiting and abdominal pain.
Please see the full Prescribing Information and Medication Guide for Mitigare® for complete product details.
NOTE: This article was not written by a medical professional and is not intended to substitute for the guidance of a physician. These are not Hikma’s recommendations for gout flare prevention, but rather facts and data collected from various reliable medical sources. For a full list of resources and their attributing links, see below.
